The Ebola Vaccine Is Highly Effective in Eastern Congo
By experts and staff
- Published
- John CampbellRalph Bunche Senior Fellow for Africa Policy Studies
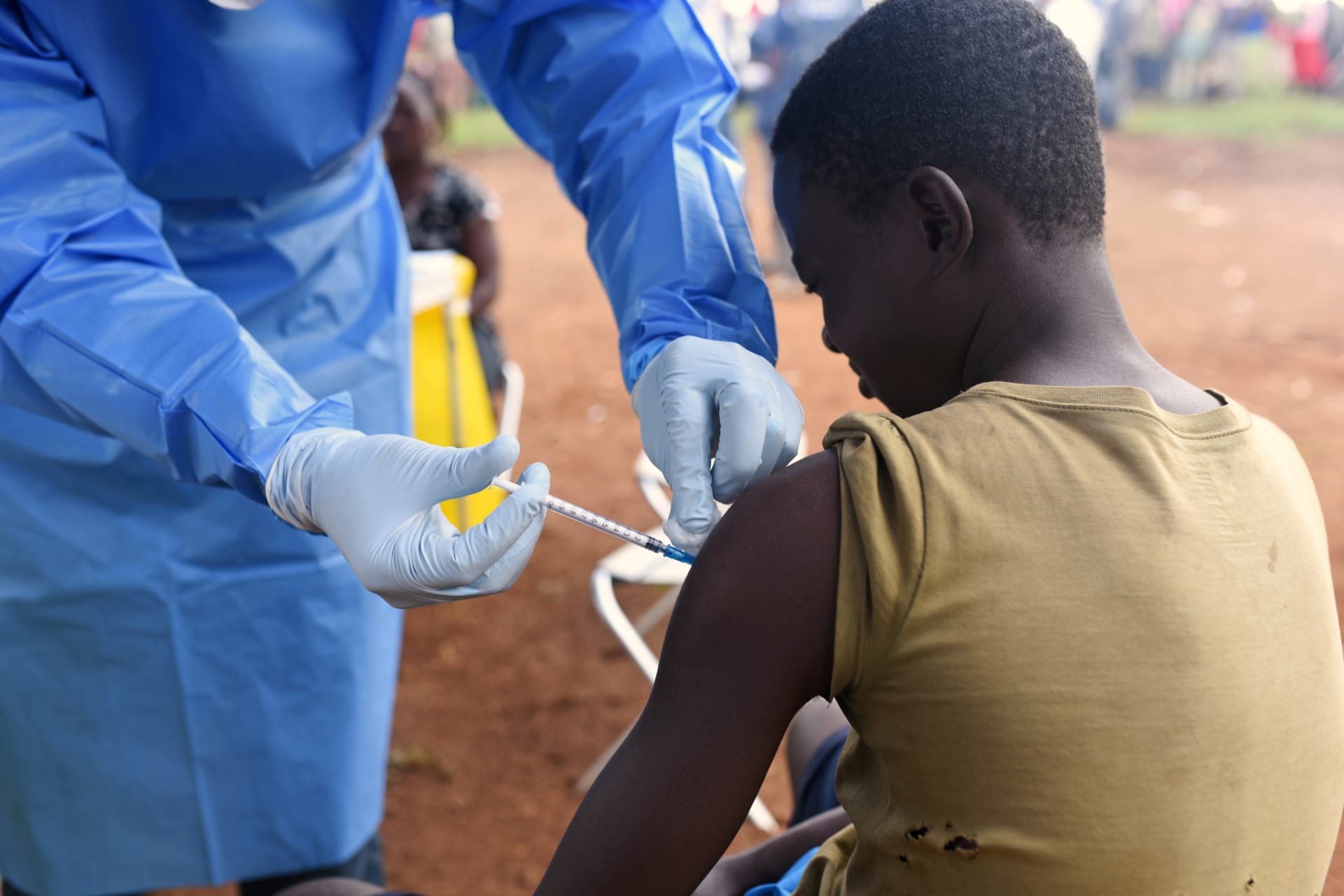
An experimental vaccine developed by Merck & Co is proving to be 97.5 percent effective at preventing Ebola. The World Health Organization’s Strategic Advisory Group of Experts had permitted the use of the vaccine based on its Expanded Access/Compassionate protocol for experimental vaccines. The vaccine is already protecting some ninety thousand people in the eastern Congo, where there has been a devastating outbreak of Ebola.
The vaccine uses a live virus rendered harmless to humans. It stimulates the human immune system to generate antibodies that attack the virus. Health workers are following the same “ring vaccination strategy” used successfully against small pox. They vaccinate those that have come into contact with the disease and also the contacts of the contacts, creating “rings of immunization.”
In the eastern Congo, still to be overcome is suspicion of health works and vaccines by local people. Militants have attacked health workers and their facilities with some popular support. Observers are calling for new strategies to overcome local opposition. The eastern Congo outbreak is now Ebola’s second largest, after the West Africa outbreak, which lasted from 2013 to 2016. Since the start of the eastern Congo outbreak in August 2018, there have been 1,198 confirmed cases and 66 probable. Of these, 748 confirmed victims have died, with an additional 66 deaths probably caused by the disease.
Ebola mortality among those not vaccinated is about fifty percent. The vaccine takes about ten days after it is administered to become fully effective. However, in eastern Congo, among those who received the vaccine and became ill before the ten-day period had elapsed, all survived.
This unalloyed good news. Now there is a medical way to vaccinate successfully against a horrible disease.