A Tale of Two Anti-Ebola Drugs
Since April, cases of Ebola virus disease (EVD) have been reported in the Democratic Republic of Congo. As of June 6, there were eight probable and confirmed cases, which were associated with four deaths. Thanks to the rapid and effective response coordinated by the World Health Organization (WHO) and its partners, the outbreak appears to have been brought under control.
That’s a huge relief. But does that mean our toolbox is indeed adequately equipped to combat the virus? What if EVD, which kills 25 to 90 percent of those who become infected, again spread like wildfire? To address these questions, the story of China’s efforts to develop anti-Ebola drugs is illustrative of the opportunities and challenges we face in ensuring global health security.
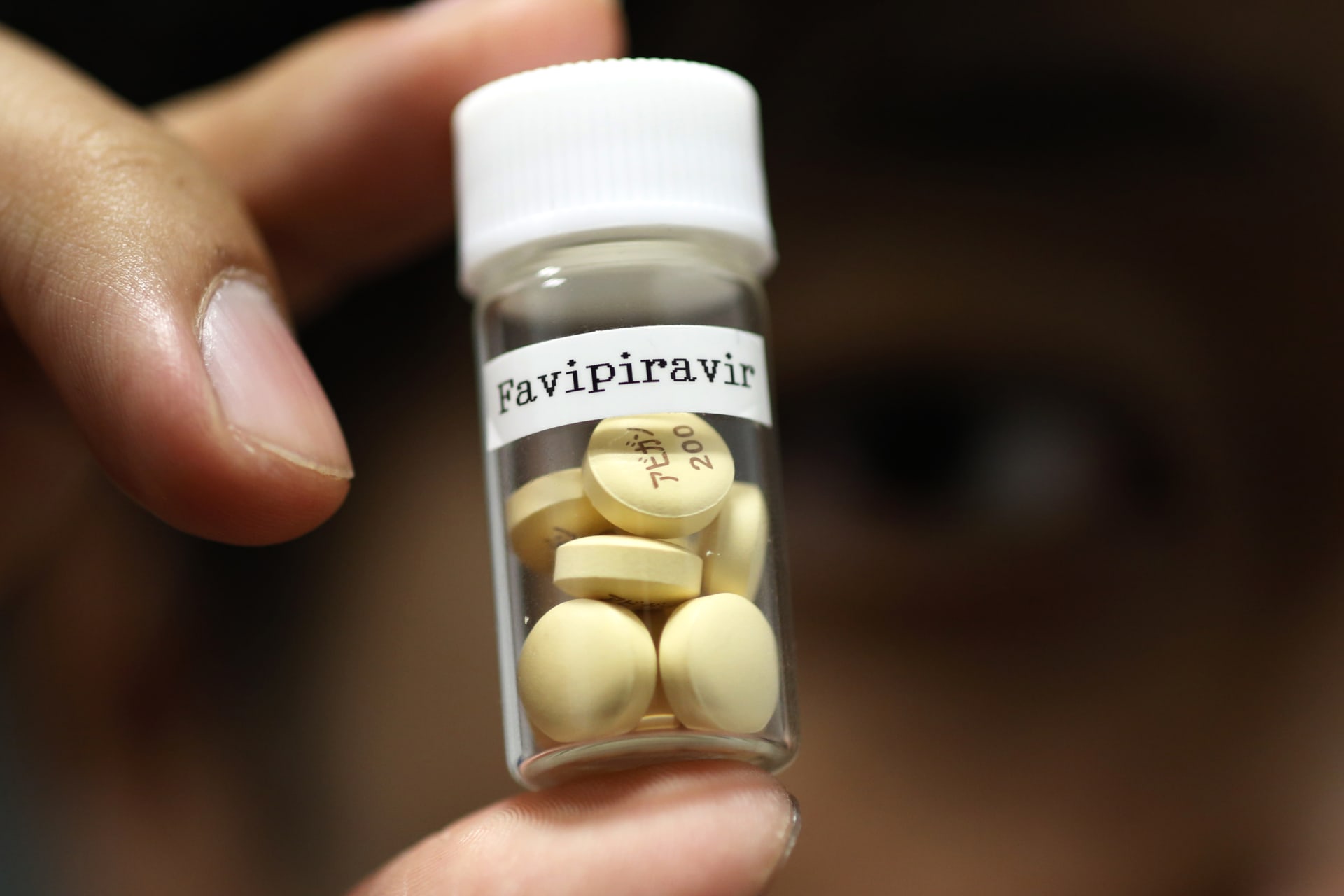
In August 2014, when Ebola virus was wreaking havoc in West Africa, the People’s Liberation Army’s (PLA’s) Academy of Military Medical Science (AMMS) announced that it had successfully developed jk-05, China’s first drug for treating EVD. The chemical formula of jk-05 remains not fully disclosed; all we know is that it is a small-molecule drug with a relatively simple structure, which should be amenable to mass production if testing proves successful. The lack of public information about the drug makes it difficult to assess its efficacy. The drug is said to have passed evaluation by PLA health experts, but it was clear that when that news came out the drug was still at an early stage of development. Not surprisingly, jk-05 was approved as a special drug for military needs and for emergency use only. That has not deterred Sihuan Pharmaceutical, originally affiliated with AMMS and now one of China’s leading generic pharmaceutical companies, from announcing in October that it had purchased the rights to commercialize jk-05 and would develop it into a broad-spectrum antiviral drug. Meanwhile, the Chinese military shipped thousands of doses of jk-05 to West Africa for treating potential infections of Chinese health workers and potentially conducting clinical trial.
But according to the WHO, jk-05 was likely a copycat product of favipiravir (T-705), an anti-influenza drug developed by Fujifilm Holdings Corp. of Japan. It was reported that the Japanese company, which registered favipiravir in China in 2006, suspected that AMMS used the patent information to develop jk-05 and demanded China to investigate this issue. As a repurposed drug, favipiravir appears to be effective against EVD in a mouse model, but its efficacy against human Ebola infection is unproved. A retrospective clinical case series was performed by Chinese scientists for EVD patients in the Sierra Leone-China Friendship Hospital in October–November 2014. It was found that the overall survival rate in the group treated with favipiravir was higher than that of the control group, but the limited sample size precluded any strong conclusions. Another subsequent and more robust clinical study by Doctors Without Borders (MSF) could not reach a conclusion about the efficacy of the drug because the number of enrolled patients was ultimately too small. Interestingly, MSF was not aware that China had used favipiravir in Sierra Leone when MSF itself was planning to test the very same drug in Guinea in December 2014. This lack of collaboration was a missed opportunity to draw strong conclusions on favipiravir, as the two projects used non-comparable methodologies.
The fate of jk-05/favipiravir highlights the role of ZMapp, an experimental drug developed by U.S.-based Mapp Biopharmaceutical, as the most promising treatment against Ebola. Access to the drug was nevertheless difficult despite the urgent need to scale up production during the West African outbreak. The drug is produced in tobacco cells whose yield is low, so a limited supply of ZMapp was quickly exhausted in the fall of 2014. At the time, scientists at AMMS and a small private Chinese company, Beijing Mabworks, used information in ZMapp’s patent to copy the active part of the drug and produce a therapeutic chimeric antibody agent, MIL-77. Even though the Chinese cocktail is similar to ZMapp in terms of the antibodies used, it is amenable to large-scale production via mammalian cell expression. With the help of the Chinese pharmaceutical firm Hisun, which had developed four mammalian cell production lines, Mabworks produced about 100 doses of MIL-77 by the end of the year. The drug was reported to have successfully treated a British military nurse who contracted Ebola while serving in Sierra Leone.
Given China’s robust bio-manufacturing capabilities and the relatively low cost of producing the drug in the country, rapid scale-up of the (then) most promising treatment against EVD seemed no longer a dream. Mabworks discussed with MSF possible donations of a dozen treatments for compassionate use. However, as the Phase 1 study of MIL-77 in healthy volunteers had not yet been conducted, export of the product from China was challenging.
There was another problem: the drug comprises three chimeric monoclonal antibodies, two of which were originally developed with support from U.S. and Canadian military research agencies. Put differently, the U.S. government and Her Majesty the Queen in Right of Canada hold patents for two of the antibodies in ZMapp. As a result, some American government officials have expressed patent infringement concerns. In 2015, Mabworks signed an agreement with Mapp Bio, the license-holder of ZMapp. Similar to China’s international marketing experience with artemisinin, Mapp Bio acquired the rights to market MIL-77 worldwide except in China. While China was able to produce thousands of doses of MIL-77 within a short period of time, Mapp Bio was not keen on further developing the Chinese biosimilar. Meanwhile, as the Ebola epidemics burned out, a clinical study on ZMapp was conducted but could not reach significant results because of the difficulty of recruiting patients (only seventy were enrolled for the study by the end of 2015). So we are back to square one: according to Julien Potet of MSF, the only ZMapp-like product that is available today is the original tobacco cell-derived ZMapp; only 100 doses of treatments are available for an Ebola outbreak.
The tale of the two Chinese-developed anti-Ebola drugs has revealed the possibilities and problems pharmaceutical industries of emerging economies face in contributing to global health security. As shown by Mabworks’ ability to rapidly produce a biosimilar of ZMapp, pharmaceutical molecularization creates opportunities for China to catch up technologically with industrialized countries in developing medical countermeasures efficiently and effectively to address major disease outbreaks. Framing infectious disease as a security challenge also enabled military actors such as AMMS, which was established to prepare for biological warfare, to play a prominent role in this process. That Chinese scientists and pharmaceutical firms could work together with their counterparts in the United States and Canada also shows the potential of North-South cooperation in the development and distribution of lifesaving drugs.
But does this suggest a paradigm shift? Is China becoming a game-changer in global health security? The fact that the development of both jk-05 and MIL-77 primarily relied on previous research done elsewhere indicates that China’s ability to offer original, innovative solutions remains in question despite having a strong pharmaceutical industry. It was challenging for China and MSF to cooperate, showing that a siloed approach continues to prevent China from working with non-state actors in drug research and development. Unfortunately, intellectual property issues remain an insurmountable hurdle for China to scale up the production of MIL-77 and serve as a real leader in this area. New norms and rules will be needed to balance public health and intellectual property considerations in pursuing and promoting global health security.
